HUREL® Human™
Co-culture comprised of cryopreserved primary human hepatocytes cultured with cells of non-parenchymal, stromal type.
HUREL® Human™
| Substrate | Enzyme | Concentration (µM) | Day 1 | Day 4 | Day 8 |
|---|---|---|---|---|---|
| Midazolam | CYP3A4 | 5 | 0.18 | 0.25 | 0.13 |
| Dextromethorphan | CYP2D6 | 20 | 0.50 | 0.25 | 0.21 |
| Tolbutamide | CYP2C9 | 20 | 0.17 | 0.14 | 0.08 |
Cryopreserved primary human hepatocytes were thawed and plated with HUREL PlatinumHeps™ Media supplemented with 10% serum and subsequently changed 24 hours post-seeding to HUREL PlatinumHeps™ Basal Media. CYP substrate concentrations are given in the table above along with metabolite formation recorded as nmoles/hr/106 cells. All incubations were carried out in triplicate on days 1, 4, and 8 upon cell delivery and incubated for 60 minutes. Reactions took place in a humidified incubator at 37°C, in 5% CO2. Collected supernatants were stored at -20°C until further LC/MS/MS analysis.
Day 1 – Morphology

Phase contrast image in a 24-well at a 10X magnification
Day 7 – Morphology

Phase contrast image in a 24-well at a 10X magnification.
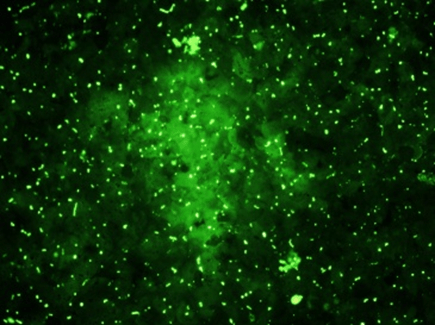
Day 7 – Bile Canaliculi
Bile canaliculi assayed via 5-(and-6)-carboxy-2′, 7′ – dichlorofluorescein diacetate (C-DCFDA) stain at a concentration of 5 microns and imaged in the GFP channel in a 96-well at 10X magnifcation with filters EX/EM 492-495/512-527 nm.
Human Donor Demographics
- Gender: Male
- Age (yrs)” 65
- Race: Caucasian
- Cause of Death: IHC-Stroke
- Drug History: Not given
- Medications: Unknown statin, Lipitor, Lisinopril, Clonaidine, and Multivitamin
- Medical History: Basel cell: 15-20 years ago. Removed with no radiation-received no chemo or radiation with no reoccurrence. Hypertension diagnosed 20-25 years ago.
Donor Serology
- Hepatitis B: Negative
- Hepatitis C: Negative
- HIV: Negative
- EBV: IgG [+], IgM [-}
- RPR: Negative
- CMV: Positive

