Reaction Phenotyping Assay
Using HURELhumanPool™ primary hepatocyte micro liver model, intrinsic clearance rates for parent drug depletion in the absence and the presence of CYP inhibitors are measured to determine the contribution of CYP enzymes. This information is useful to identify the specific enzymes responsible for the drug metabolism.
Background
- Reaction phenotyping studies (i.e., drug-metabolizing enzyme identification studies) are important in the drug discovery and development processes as it provides useful information for predicting clearance pathways, victim drug-drug interaction, and potential variability in drug pharmacokinetics.
- The FDA recommends evaluating major CYP enzymes (CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP3A) using in vitro reaction phenotyping experiments to determine which enzymes metabolize the investigational drug [1].
- Current in vitro reaction phenotyping systems using human recombinant enzymes and liver microsomes have some drawbacks in terms of physiological relevancy, incomplete enzymes (e.g. SULT, non-CYPs) and cofactors, cost-effectiveness. These models are specifically not applicable for low-clearance compounds in the detection of drug depletion [2].
- Visikol utilizes the HURELhumanPool™ Primary Hepatocyte Micro Liver Model, which maintains stable metabolic activities in long-term cultures (> 4 weeks). This model is more physiologically relevant and has a complete complement of drug metabolizing enzymes (CYPs and non-CYPs) and cofactors.
- Using HURELhumanPool™ primary hepatocyte micro liver model, intrinsic clearance rates for parent drug depletion in the absence and the presence of CYP inhibitors are measured to determine the contribution of CYP enzymes. This information is useful to identify the specific enzymes responsible for the drug metabolism.
General Procedure
- HURELhumanPool™ primary hepatocyte micro liver model is cultured for one week prior to dosing compounds.
- Treatment with test compounds in the absence and the presence of CYP inhibitors.
- Repeated dosing of CYP inhibitors every 24 hours.
- At each time point, samples are collected for LC-MS/MS analysis.
- Intrinsic clearance is calculated using the depletion of the parent compound.
| Instrument | LC-MS/MS system composed of an Agilent 1100 LC coupled with a SCIEX QTRAP 4000 | |||||||||||||||||||||||||||
| Analysis Method | LC-MS/MS | |||||||||||||||||||||||||||
| Positive Controls and Inhibitors | 7 Major CYP Enzyme Control Panel (recommended by FDA):
|
|||||||||||||||||||||||||||
| Cell model | HURELhumanPool™ Primary Hepatocyte Micro Liver Model
(Primary human hepatocytes (5 donors) co-culture with stromal cells) |
|||||||||||||||||||||||||||
| Time points | t = 0, 24, 48 hours after dosing (custom time points available) |
|||||||||||||||||||||||||||
| Test Article Concentration | Single point assay (1 µM) (custom concentrations available) |
|||||||||||||||||||||||||||
| Number of Replicates | 3 replicates per time condition | |||||||||||||||||||||||||||
| Test Article Requirements | 50 uL of 20 mM solution or equivalent amount of solid | |||||||||||||||||||||||||||
| Data Delivery | % remaining of parent compounds at each time point
Half-life (t½) Intrinsic clearance (CLint) CYP contribution (fm) |
Validation Data
Control Compounds
Using HURELhumanPool™ Primary Hepatocyte Micro Liver Model, major CYP enzyme and inhibition functions for probe substrates were evaluated. Depletion of parent substrates were analyzed by LC-MS/MS. We have demonstrated that clearance rates for control substrates in the presence of inhibitors are significantly decreased. The results indicate that the HUREL model can be used for reaction phenotyping studies to determine drug clearance pathways and the contribution of drug metabolizing enzymes.
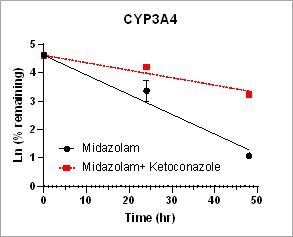
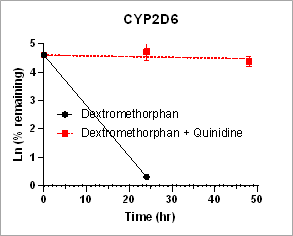

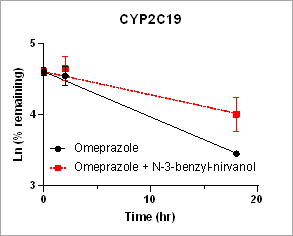
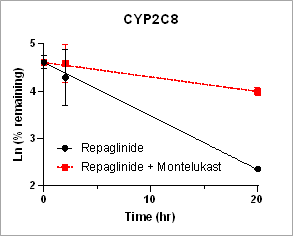
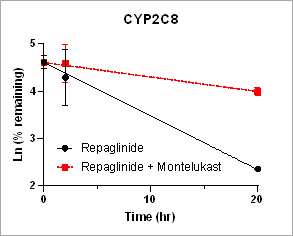
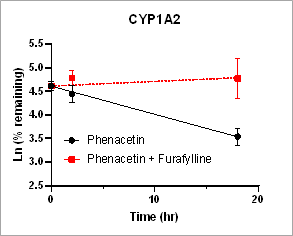
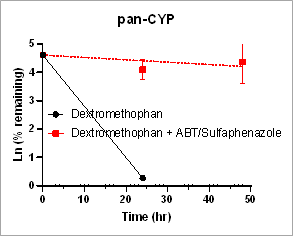
Figure 1. Clearance plots for control substrates in the absence and in the presence of inhibitors in HURELhumanPool™ Primary Hepatocyte Micro Liver Model. Pan-CYP inhibitors are used to determine the contribution of CYP vs. non-CYP enzymes. Data are expressed as mean ± SEM.
References
- FDA Guidance for Industry – In Vitro Drug Interaction Studies – Cytochrome P450 Enzyme- and Transporter-Mediated Drug Interactions (January 2020)
- Di, L. (2017). Reaction phenotyping to assess victim drug-drug interaction risks. Expert opinion on drug discovery.

