Skin Brightening
The MelanoDerm Skin Brightening Assay provides invaluable in vitro data as an early screening tool for raw materials to reduce costs and increase the chances of a formulation’s success prior to clinical trials. Cosmetics manufacturers continually develop safer and more effective skin brightening products for their customers struggling with melasma and post-inflammatory hyperpigmentation. Evaluate the efficacy of cosmetic and/or pharmaceutical skin care formulations that are used to combat skin pigmentation disorders with MelanoDerm.
Mattek offers MelanoDerm Skin Brightening/melanogenesis assay as a non-GLP service.
Protocol
| Test Model | MelanoDerm |
| Replicates | N=8 tissues per test condition |
| Exposure Conditions | 3 treatments/week for 2 weeks with 25µl of test material and controls |
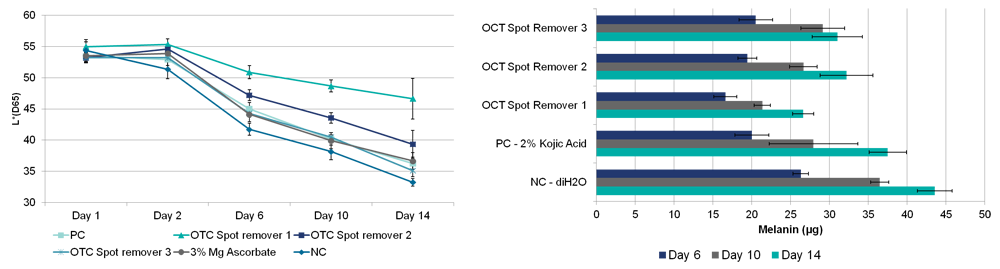
| Assay Controls | 2% Kojic Acid (positive control), DI H2O (negative control) |
| Endpoints | Macroscopic analysis, Histological analysis, Quantitative Melanin Assay |
| Data Delivery | Histological images and analysis, Melanin Assay Analysis, Macroscopic photos skin brightening potential |
Data

Request a Quote
Thank you for requesting information about Mattek products! A representative will contact you shortly.
**If you would like to place an order for Mattek products, please contact Customer Service**


