EpiCorneal™
The EpiCorneal 3D human tissue model provides a highly predictive non-animal alternative to assess ophthalmic drug delivery, wound healing and tissue regeneration, disease modeling (e.g. dry eye) and corneal infection.
EpiCorneal™
The EpiCorneal 3D human tissue model provides a highly predictive non-animal alternative to assess drug delivery, infection, wound healing/tissue regeneration and disease modeling (i.e. Dry Eye).
Download the EpiCorneal Data Sheet
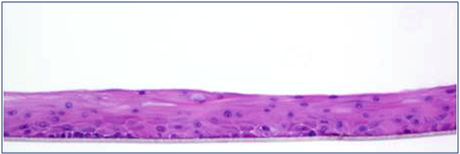
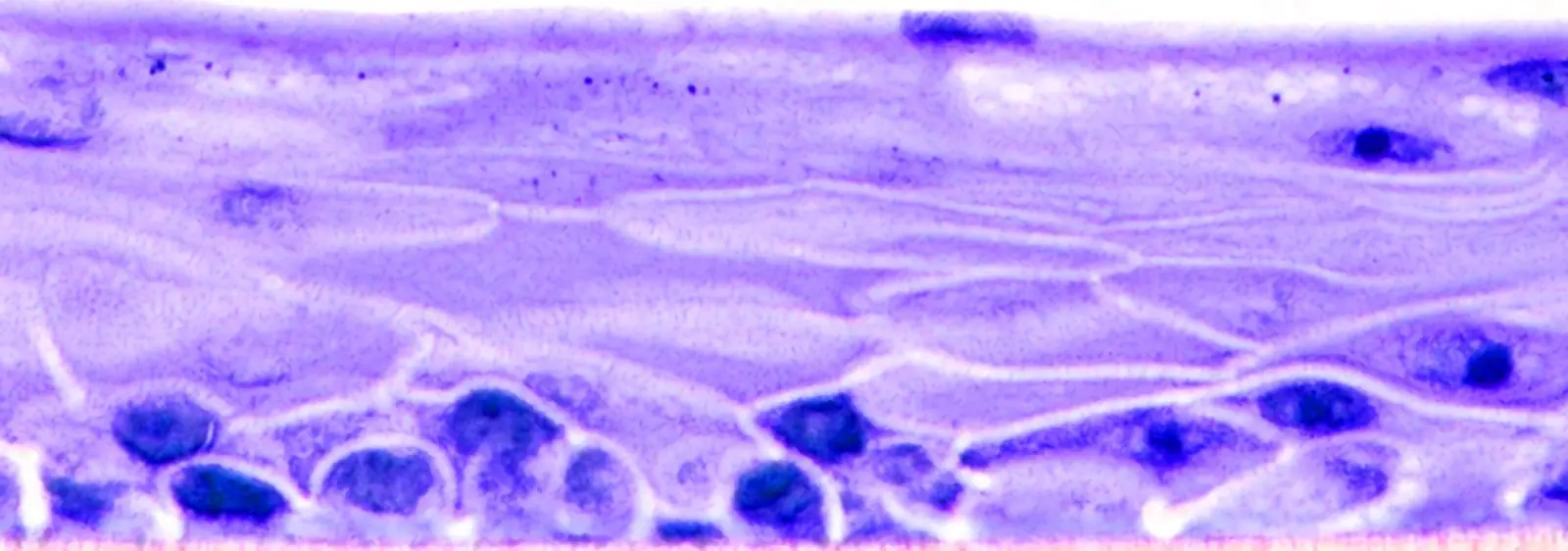
Mattek’s EpiCorneal tissue model consists of normal human corneal epithelial cells cultured to form a stratified, squamous epithelium which closely parallels normal human corneal tissue. The corneal cells, cultured on specially prepared cell culture inserts with serum-free medium, differentiate to form a multi-layered structure.
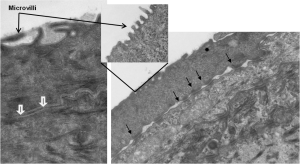
TEM Analysis of EpiCorneal illustrates the presence of 1) microvilli, 2) tight junctions (white arrows), and 3) desmosomes (black arrows)
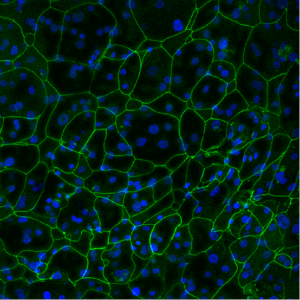
Used in drug delivery and drug development, EpiCorneal is a mitotically and metabolically active tissue with barrier properties approaching those of the human cornea. EpiCorneal can be stimulated to release many cytokines known to be important in inflammation and diseases such as dry eye. Further, the advanced 3D tissue model expresses cornea specific drug transporters and metabolizing enzymes.
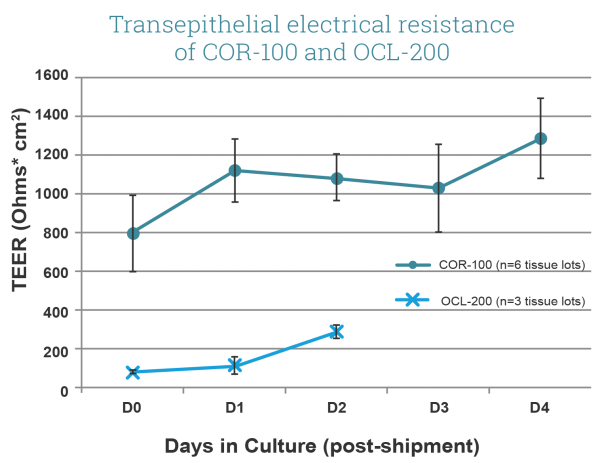
Transepithelial Electrical Resistance (TEER) of EpiCorneal (COR-100, comprised of corneal epithelial cells) and EpiOcular (OCL-200, comprised of keratinocytes) tissue models. Note high TEER of COR-100 (avg. > 600 Ω*cm2) when compared to OCL-200 (~ 200 Ω*cm2). Culture area is 0.6 cm2.
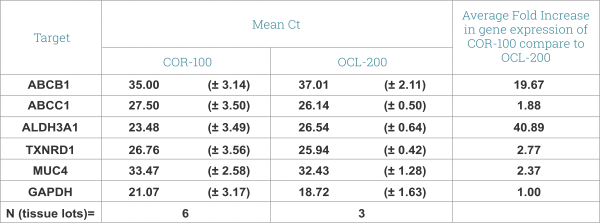
Expression of drug transporters and cornea-specific markers in EpiCorneal. qPRC data shows expression of ABCB1(p-gp) and ABCC1 (CFTR/MRP) – ATP-binding cassette, efflux transporters, multidrug resistance proteins with important role in drug disposition and distribution. Aldehyde Dehydrogenase 3 (ALDH3-A1) promotes resistance to UV and 4-hydroxy-2-nonenal-induced oxidative damage in the cornea. Pyridine nucleotide oxidoreductase (TXNRD1 or TrxR1) protects against oxidative stress. MUC4 – Mucin 4 is found predominantly in the most superficial cell layers in stratified corneal epithelium.
Corneal Drug Delivery and Corneal Toxicity
EpiCorneal closely recapitulates the physiology, 3D tissue architecture, and function of the human cornea for use in pharmaceutical development applications.
Latanoprost Drug Delivery
Model Compound Drug Delivery
Dry Eye Disease
EpiCorneal Dry Eye Disease Application Note
Corneal Wound Healing
EpiCorneal Wound Healing Application Note
Browse our reference library to see how our EpiCorneal tissue has been used in these areas of study.
Tissue
Kit: A standard EpiCorneal kit (COR-100) consists of 24 tissues. (Tissue “kits” contain tissues, a small amount of culture medium, and plasticware; contact Mattek for specific kit contents)
Formats: 9mm individual inserts – tissue culture substrate is chemically modified with a pore size of 0.4 μm
Culture: Air-liquid interface
Histology: 4-5 cell layers – stratified, squamous morphology and tissue structure
Lot numbers: Tissue lots produced each week are assigned a specific lot number. A letter of the alphabet is appended to the end of the lot number to differentiate between individual kits within a given lot of tissues. All tissue kits within a lot are identical in regards to cells, medium, handling, culture conditions, etc.
Shipment: At 4°C on medium-supplemented, agarose gels
Shipment day: Every other week
Delivery: Tuesday morning via FedEx priority service (US). Outside US: Tuesday-Thursday depending on location
Shelf life: Including time in transit, tissues may be stored at 4°C for up to 3 days prior to use. However, extended storage periods are not recommended unless necessary. In addition, the best reproducibility will be obtained if tissues are used consistently on the same day, e.g. Tuesday afternoon or following overnight storage at 4°C (Wednesday morning)
Cells
Type: Normal human corneal epithelial cells (HCEC)
Genetic make-up: Single donor
Derived from: Healthy donor
Screened for: HIV, Hepatitis-B, Hepatitis-C, mycoplasma
Medium
Base medium: Dulbecco’s Modified Eagle’s Medium (DMEM).
Growth factors/hormones: Epidermal growth factor, insulin, hydrocortisone and other proprietary stimulators of corneal epithelial differentiation.
Serum: None.
Antibiotics: Penicillin (50 U/ml) / Streptomycin (50 µg/ml)
Anti-fungal agent: Amphotericin B (0.125 µg/ml).
pH Indicator: Phenol red.
Alternatives: Phenol red-free, antibiotic/anti-fungal-free, or hydrocortisone-free medium and tissue are available. Agents are removed at least 3 days prior to shipment.
Maintenance medium: Most experiments with EpiCorneal™ are performed within 48 hours. However, for longer experiments, the tissue can be maintained in COR-100-MM (identical to EpiCorneal™ assay medium).
Quality Control and Sterility
Visual inspection: All tissues are visually inspected and if physical imperfections are noted, tissues are rejected for shipment
Sterility: All media used throughout the production process is checked for sterility. Maintenance medium is incubated with and without antibiotics for 1 week and checked for sterility. The agarose gel from the 24-well plate used for shipping is also incubated for 1 week and checked for any sign of contamination
Screening for pathogens: All cells are screened and are negative for HIV, hepatitis B and hepatitis C using PCR. However, no known test method can offer complete assurance that the cells are pathogen free. Thus, these products and all human derived products should be handled at BSL-2 levels (biosafety level 2) or higher as recommended in the CDC-NIH manual, “Biosafety in microbiological and biomedical laboratories,” 1998. For further assistance, please contact your site Safety Officer or Mattek technical service
Notification of lot failure: If a tissue lot fails our QC or sterility testing, the customer will be notified and the tissues will be replaced without charge when appropriate. Because our QC and sterility testing is done post-shipment, notification will be made as soon as possible (Under normal circumstances, ET-50 failures will be notified by Wednesday 5 p.m.; sterility failures will be notified within 8 days of shipment)

